Safety Document Distribution Made Simple
A single, automated platform for the oversight and distribution of Safety content.
LifeSphere® Technology is trusted by these industry leaders:







Document Distribution
Document Distribution simplifies your global drug safety and safety reporting operations by making it quick and easy to distribute safety reports to key stakeholder groups.
Audit Readiness
Enable improved oversight and greater ease maintaining compliance with regulators.
Maximize Efficiency
Automate manual processes to enable the distribution of safety reports in real-time.
Enhanced Collaboration
Achieve greater cross-functional transparency via a shared platform and reporting.
Cost Savings
Save time and effort with out-of-the-box and regularly updated reference models.
Features
Deliver real value with Document Distribution.

Secure SaaS Platform
Provide a secure, web-based platform for users to centrally manage safety content, seamlessly integrated with your safety platform.

Content Binders
Support audit readiness with out-of-the-box binder structures with template placeholders.

Next-Gen Tracking
Facilitate the tracking of distributions through flexible and customizable dashboards and comprehensive audit trails for documents.

Reduce Site Burden
Streamline cross-reporting and prevent duplicate distributions with a single point of access for all portal users to view relevant distributions.

Enhanced Training Module
Facilitate the creation, scheduling, publishing, and assignment of training for specific targeted users or contacts, while tracking compliance through reports.

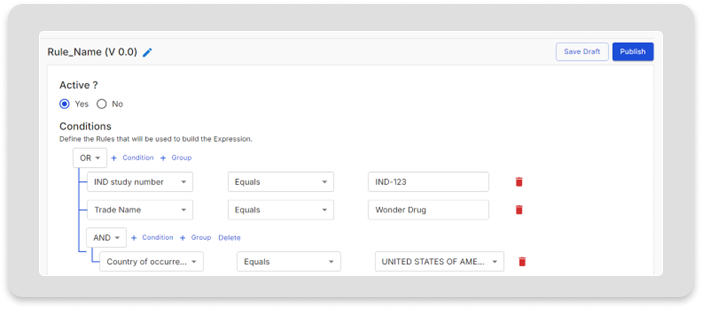
Advanced Rule Builder
Enable the creation of customized rules at product, study, site, and country levels to comply with varying regulations across different countries.
Learn more about our collaborative partnership with customers.
"*" indicates required fields

ArisGlobal is a pivotal partner in the life sciences industry, specializing in solutions that drive drug development, safety monitoring, and regulatory compliance. We collaborate with more than 500 global life sciences companies, CROs, and government health authorities, including leading biopharmaceutical firms and regulatory bodies such as FDA, Health Canada, and NMPA.
Headquartered in the United States, ArisGlobal has regional offices in Europe, India, Japan and China.
Quick Links
© 2024 ArisGlobal – All Rights Reserved
