Analytics
Gain enhanced insights and enhance decision-making across your regulatory operations.
LifeSphere® Technology is trusted by these industry leaders:







Analytics
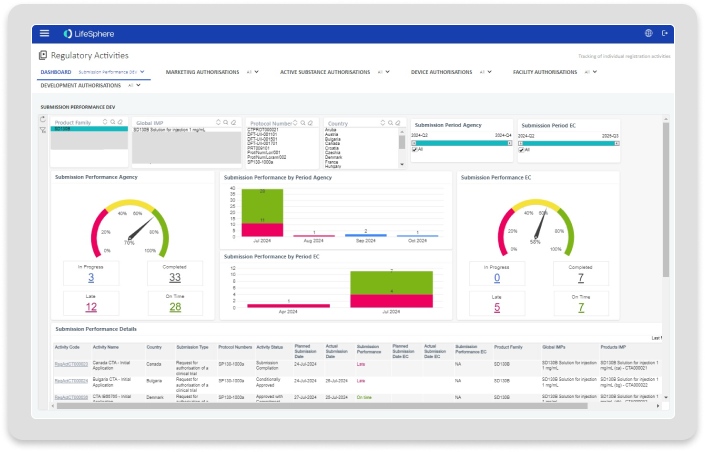
Analytics provides a preconfigured set of standard reports addressing common tracking, reporting and analytical needs across the regulatory lifecycle.
Robust Analytics
Leverage advanced analytics tools to ensure users get deeper insights and can make more informed decisions.
Accessible Insights
Give users rapid and easy access to critical insights without needing extensive technical expertise or system knowledge.
Enhanced Visualization
Present insights as needed and where needed with seamlessly embeddable and interactive dashboards.
Centralized Insights
Establish a single source of insights for regulatory compliance and business performance.
Features
Deliver real value with Analytics.

Deeper Insights
Leverage robust analytics tools to derive advanced metrics.

Self-Service Tools
Create custom or personalized reports using an easy to use report editor.

Embedded Analytics
Seamlessly embedded reports and dashboards to present data as needed.

Information Security
Control permissions and restrict information access to specific users.

Insight Sharing
Easily collaborate and share reports with other users.
Related Resources
Learn more about our collaborative partnership with customers.
"*" indicates required fields

ArisGlobal is a pivotal partner in the life sciences industry, specializing in solutions that drive drug development, safety monitoring, and regulatory compliance. We collaborate with more than 500 global life sciences companies, CROs, and government health authorities, including leading biopharmaceutical firms and regulatory bodies such as FDA, Health Canada, and NMPA.
Headquartered in the United States, ArisGlobal has regional offices in Europe, India, Japan and China.
Quick Links
© 2024 ArisGlobal – All Rights Reserved
